flu360 resources help streamline and strengthen immunization clinics year-round, putting more into flu vaccine administration than just the flu shot ingredients.
flu360 Solutions
Need support across your business and throughout the year? We have solutions.


Clinical Support


Financial Guidance
flu360 makes it easy and expedient to prioritize your patients and influenza vaccinations without putting off ordering, coding, billing, and reimbursements.


Operational Efficiency
flu360 simplifies the complex, providing greater efficiencies to meet day-to-day operational burdens and inventory management.
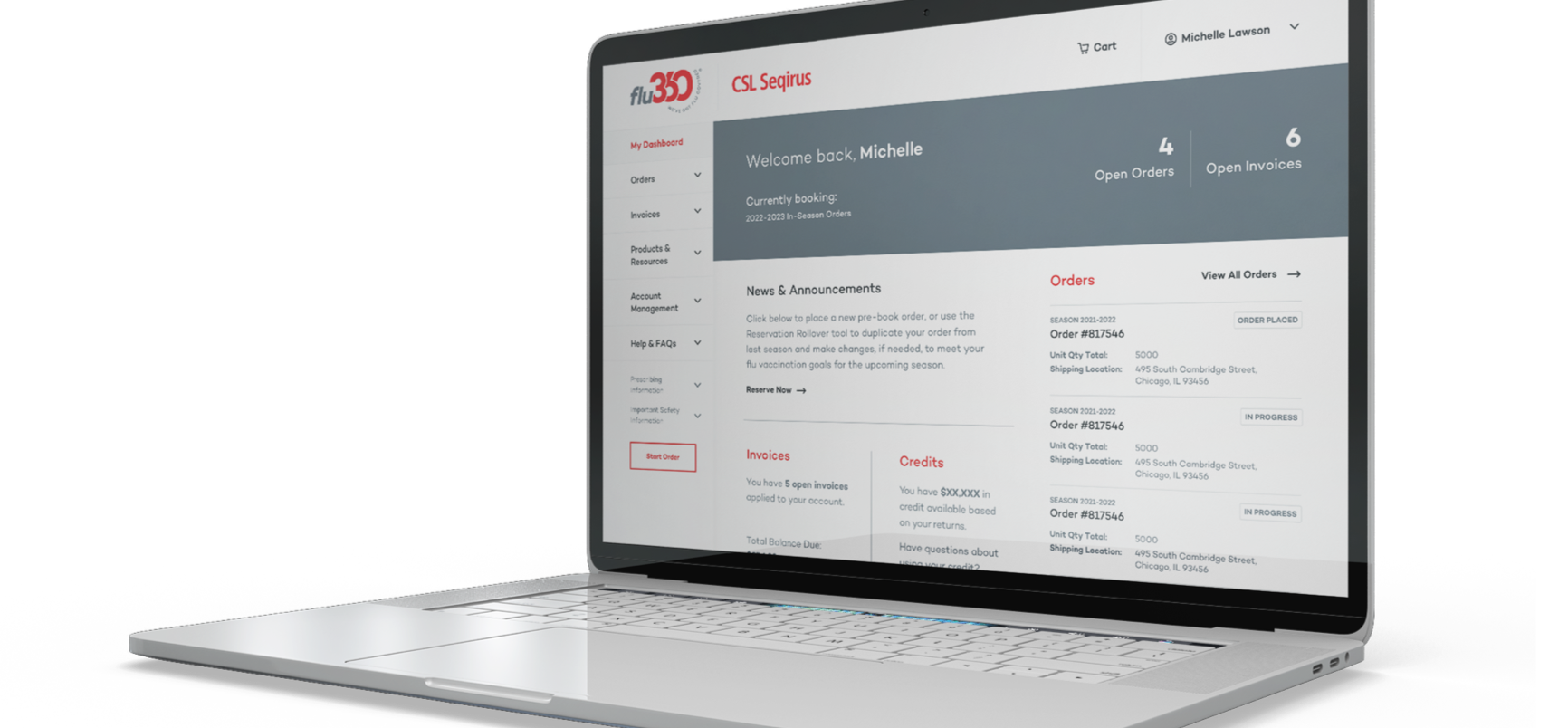
Preview the possibilities of the flu360 dashboard
Create an account for free, and access the advantages of influenza vaccine support, all in one place.
- Clinical expertise, flu campaigns, and innovations
- Financial management and payment options
- Operational ordering, logistics, and support
Multiple needs call for multiple solutions, across
your business and throughout the year
What if...
you could manage immunization clinics with greater simplicity, maximizing flu vaccinations while minimizing effort?
It's possible.
Explore clinical support
What if...
you could drive financial value across your immunization business through greater efficiencies?
It's possible.
Explore financial guidance
What if...
your day-to-day vaccine operations aligned with your long-term business objectives?
It's possible.
Explore operational efficiency